
Alkaline batteries can typically be charged several dozen times without losing significant capacity. An ideal charging circuit will supervise cell temperature while charging. The termination voltage should not exceed 1.5 Volts per cell. An alkaline cell can be safely charged with a constant current source of 100mA or more. 
When recharging an alkaline cell one must take into account 3 factors: Charging current, termination voltage and cell temperature. Recharging of alkaline batteries is uncommon, but possible. However, alkaline batteries cost significantly less. 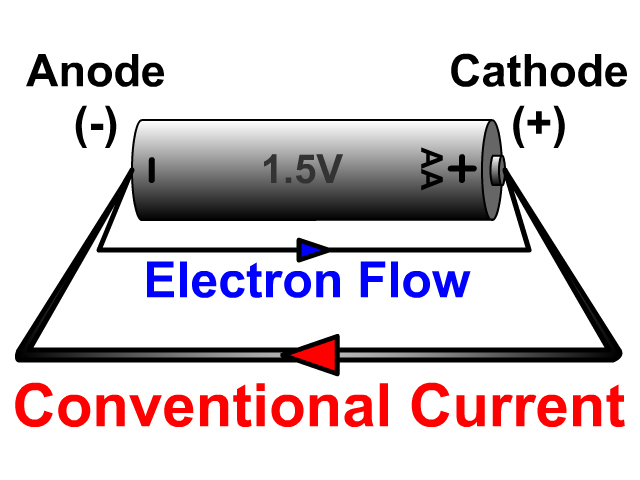
Volume for volume, alkaline batteries have inferior current handling capacity when compared to other chemistries like NiCd and NiMH. Applications requiring high currents of several amps, such as high powered flashlights and boom-boxes, will require D sized cells to handle the increased load. Larger cells, such as C and D cells, can deliver more current. A general rule of thumb is that an AA alkaline battery can deliver 1000mA without any significant heating. This is a result of decreasing internal resistance as the internal surface area of the cell increases. The amount of current an alkaline battery can deliver is roughly proportional to its physical size. A AA-sized alkaline battery might have an effective capacity of 3000 mAh at low power, but at a load of 1000 mA, which is common for digital cameras, the capacity could be as little as 700 mAh. Therefore, the capacity of an alkaline battery is strongly dependent on the load, even at moderate loads. Alkalines have a high internal resistance, and a high thermal coefficient of resistivity - the faster an alkaline battery is drained, the higher percentage of the load it dissipates as heat. Unlike NiMH rechargeable batteries, alkaline batteries are normally not sold with a nominal capacity. So far, they are comparable to zinc-carbon batteries, but the difference is that alkaline batteries use potassium hydroxide (KOH) as an electrolyte rather than ammonium chloride or zinc chloride. When describing standard "AAA", "AA", "C", "sub-C" and "D" type cells, the anode is the flat end while the cathode is the end with the raised button. In an alkaline battery, the anode (negative terminal) is made of zinc powder (allows more surface area for increased rate of reaction therefore increased electron flow) and the cathode (positive terminal) is composed of manganese dioxide.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
